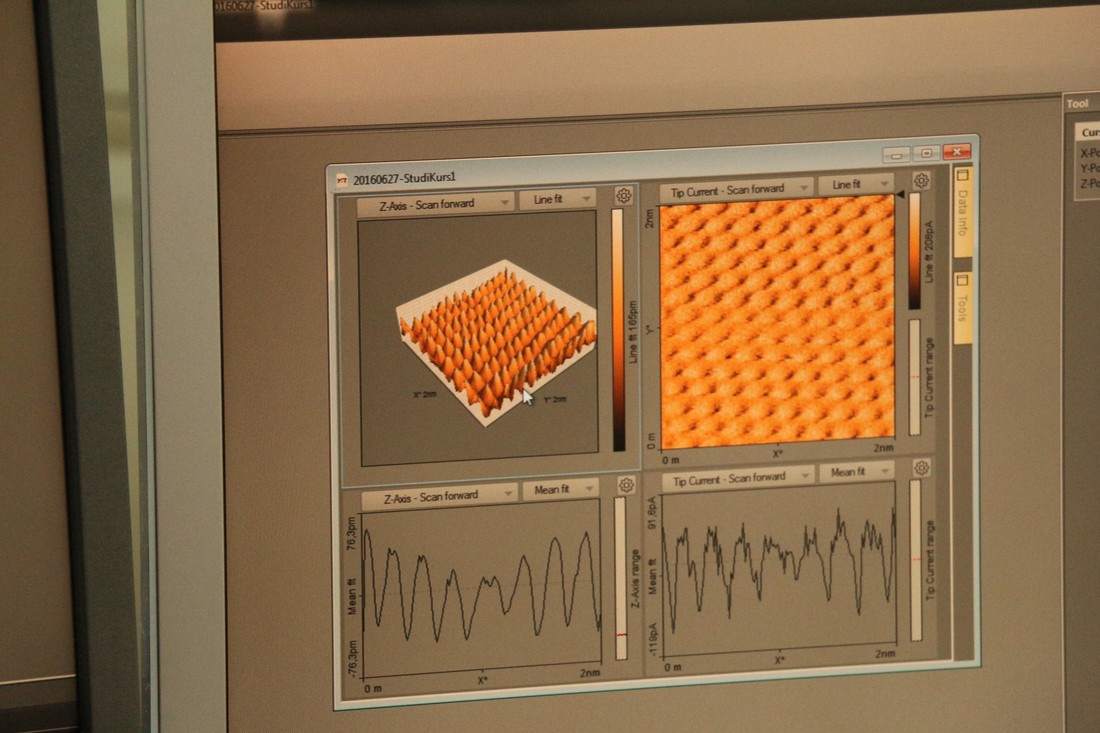
by JosephToday, we went to the Deutsches museum for a course about nanotechnology. It was our third visit to the museum. Our teacher, Andrea, who is a lecturer at TUM, first showed us silicon dioxide particles in different sizes. There was a plastic bottle with some 3mm SO2 and another plastic bottle with 7nm SO2. She shook the bottle with 3mm SO2, but nothing happened. Then, she shook the bottle with 7nm SO2; the nanoparticles acted like a liquid and their volume increased. After a while, they lost their liquid characteristics and reverted back to their original volume. It was very interesting for me because I had never seen a material behave this way before. Next, Andrea started to explain how nanoparticles could affect our daily lives. Many nanoparticles are very small, much smaller than the spaces between the molecules that make up the outer membranes of our cells. so they can get into our cells easily. Once inside the cells, they can damage the enzymes there and this can harm the systems of our bodies. Therefore, she suggested to us not to eat too much food containing nanoparticles. In the second part of our 4 hour lesson, Andrea gave each of us two tasks. One was to use a scanning tunneling microscope to see the array of atoms on the surface of a mystery substance and the other one was to look at a picture of an atomic lattice and to determine what it was. At first, Luke and I did the microscope task. We took turns to make a platinum-iridium conducting tip (one atom wide) and fix it in place inside the microscope. Luke made his tip easily, but there was something wrong with the microscope and it wouldn't register a clear image. Andrea didn't even know why this would happen. Then, it was my turn to make the tip. I tried very hard with my shaking hands and my nearsighted eyes. I failed twice and dropped both pins, but on the third attempt, I managed to do the task. After my success, we still couldn't calibrate the sensitive machine very well, so we couldn't really form a clear picture of the atoms. Next, Luke and I used a computer to do the second task. At first, we searched for tables of atomic spacing that could show us information about the substance we were observing. We calculated the length of the spaces between the atoms in nanometers and compared it to the table. The most similar data on the internet with our calculations was carbon. We told Andrea we were looking at carbon atoms, but she said it was actually graphite (a kind of form of carbon). This was definitely the most unusual and surprising course that I have ever attended.
0 Comments
Leave a Reply. |
STEMEducation
科學、技術、工程、數學 Archives
February 2018
Categories |






 RSS Feed
RSS Feed
